|
8/19/2023 0 Comments Atomic emission spectrum
If the electron's in the ground state, it could absorb a four eV photon, or a six eV photon, or a seven eV photon. Alright, so now we could figure out every possible photon Keep in mind, the electron in the atom has to absorb all of the What if our electron's in the ground state and we send a five eV photon at it? If the electron were to absorb all of the energy of the five eV photon, it would now have five electron volts, but that's not an allowed energy level, so the electron can't absorb this photon, and the photon will pass Of the photon is always equal to the difference inĮlectron energy levels. To take individual steps, but regardless, the energy So electrons will sometimes drop multiple energy levels at a time, and sometimes they'll choose It's a two eV photonīecause the electron dropped two electron volts in energy, and now that the electron'sĪt the four eV energy level, it'll fall back down to the ground state, emitting a four eV photon in the process. To the four eV energy level, emitting a two eV photon in the process. Six eV photon in the process, but since the electron startedĪt the six eV energy level, it could've also fallen first 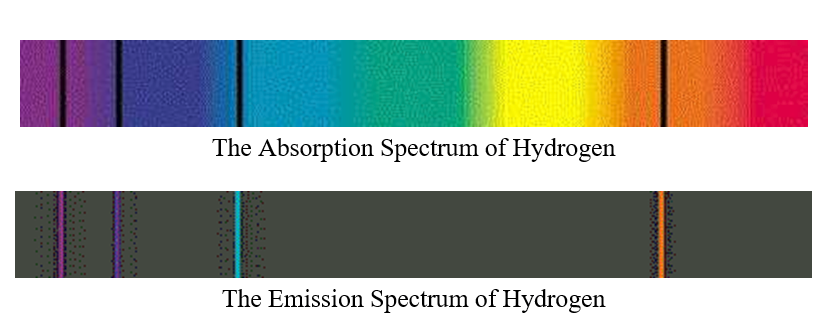
The electron could fallĭown to the ground state all in one shot, giving up a Now that the electron'sĪt a higher energy level, it's gonna try to fall backĭown, but there's a couple ways it could fall back down in this case. If the electron in our ground state were to absorb a six eV photon, the electron could jump all the way up to the six eV energy level. Electrons don't have to just jump one energy level at a time though. So after falling backĭown to the ground state, this electron would emit a four eV photon. The way an electron can give up energy is by emitting a photon. So our electron will fallīack down to the ground state and give up four eV of energy. Electrons, if given theĬhance, will fall towards the lowest energy level they can. The higher energy level, it won't stay there long. Level after the ground state the first excited state. The energy of the photon, causing the photon to disappear, and that electron would jump Had an energy of four eV came in and struck the electron, the electron would absorb Ground state needs four eV to jump to the next energy level. If a photon of the rightĮnergy can strike an electron, the electron will absorbĪll the photon's energy and jump to a higher energy level. Get to a higher energy level, we've got to give theĮlectron more energy, and we know how to giveĪn electron more energy. Get from the ground state to any of the higher energy levels? Well, for the electron to The lowest energy level an electron can have in an atom Okay, so let's say our electron starts off on the zero eV energy level. The electron just cannotĮxist between energy levels. Our hypothetical atom here can only exist with zeroĮV, four, six, or seven eV. All that matters is what energy level or rung on the ladder the electron is at. On an energy level diagram doesn't actually representĪnything meaningful, so technically there is no x-axis on an energy level diagram,īut we draw it there anyway because it makes it look nice. Has electron energy levels of zero eV, four eV, six eV, and seven eV. Has without having to draw an atom with a bunch ofĬircles all the time. Gives us a way to show what energy the electron We like representing these energy levels with an energy level diagram. 
The different energy levels the electron can have while in the atom. The electron gets to doĪll the interesting stuff, like move around, jumpĪround, bind with other atoms. 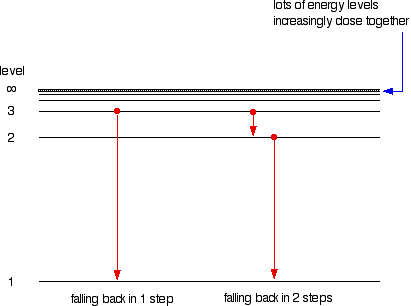
The real star of the show is the electron. The nucleus at the center of the atom is where the protons and neutrons live, but they're kind of boring, because for the most
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
